Quality Management made as Simple as ABC
Meet Effivity QMS-the most advanced, adaptable, 💸budget-friendly and 🫱🏻🫲🏽collaborative tool for
optimizing quality management.
Streamline and automate business processes with our dynamic QMS
software that evolves with your needs.







Pre-configured and highly result-oriented, Effivity QMS is truly a quality software for achieving excellence in quality management system.
Trusted by 3000+ happy customers who are leading the world









It’s not just Software.
But the future of Quality Management
With a no-code/low-code approach, Effivity QMS makes quality cycles 100% effortless,
paperless and errorless
Our interface adopts a drag-n-drop approach backed by strong visuals for instant visibility and flexibility. Endless customization capabilities make it a breeze to
Create &
Customize Forms
Fields
Workflows
Reports
and everything in between.
With Effivity, you take control. Not burden.
A Modern QMS for Modern Needs
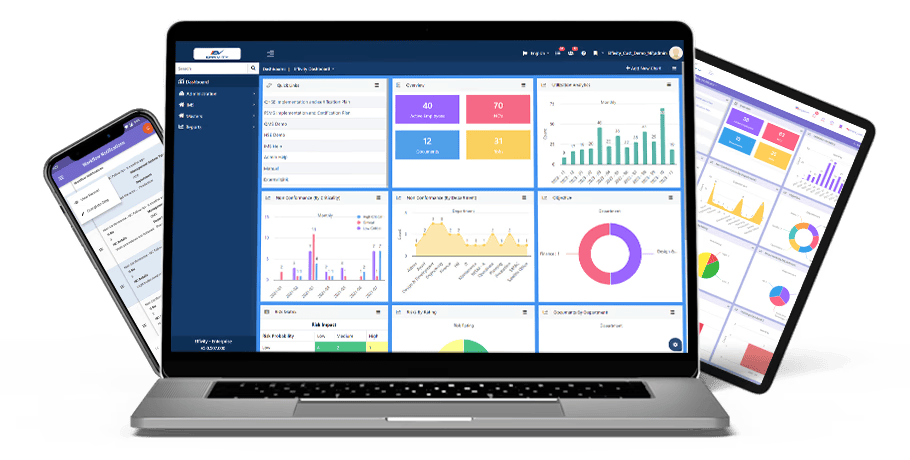
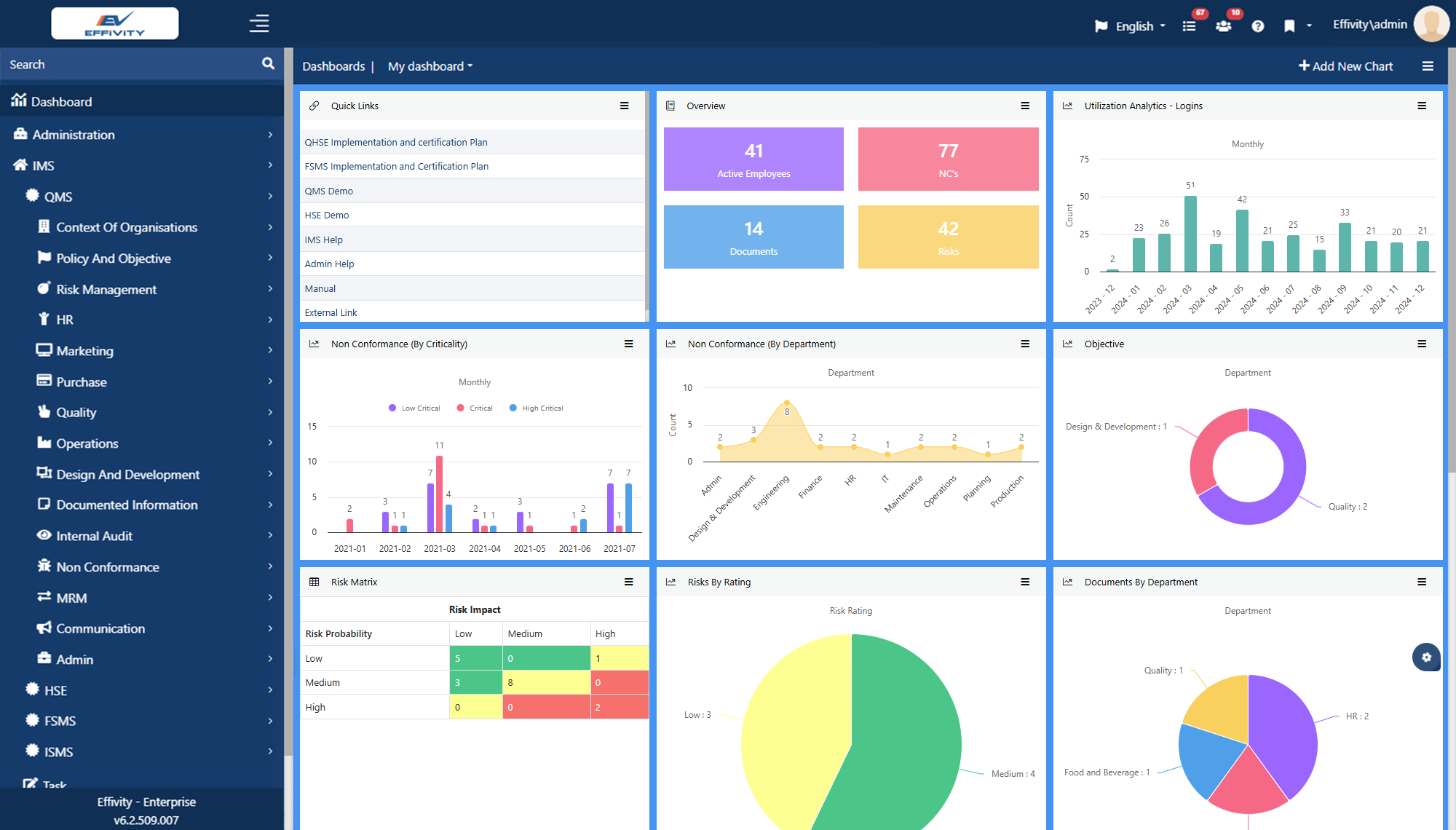
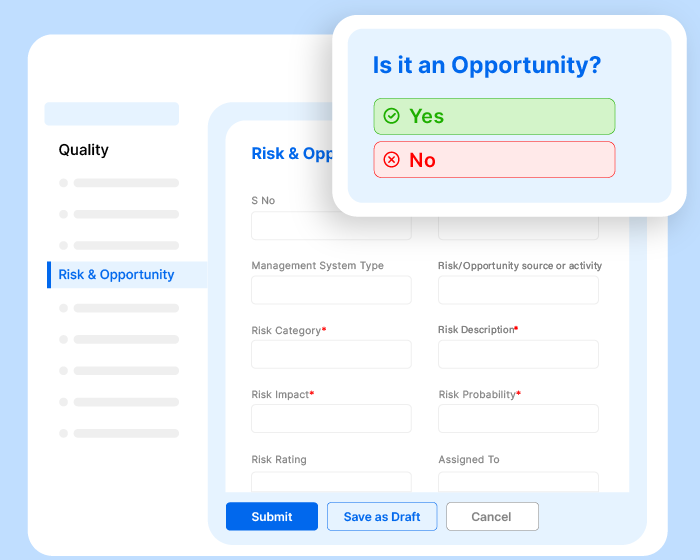
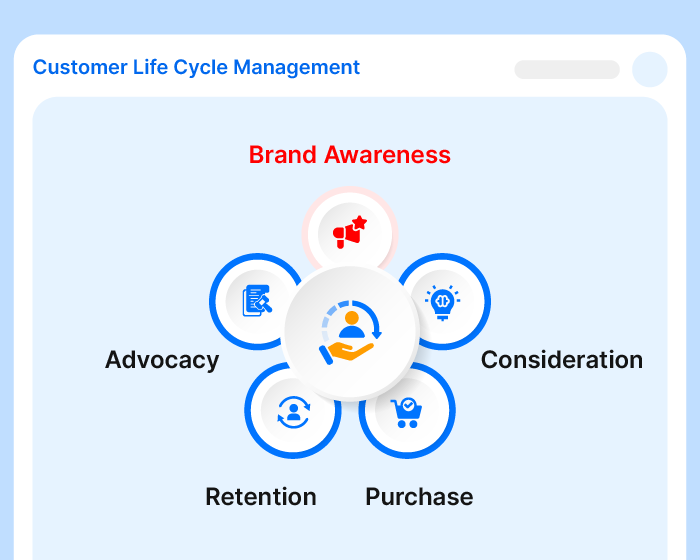
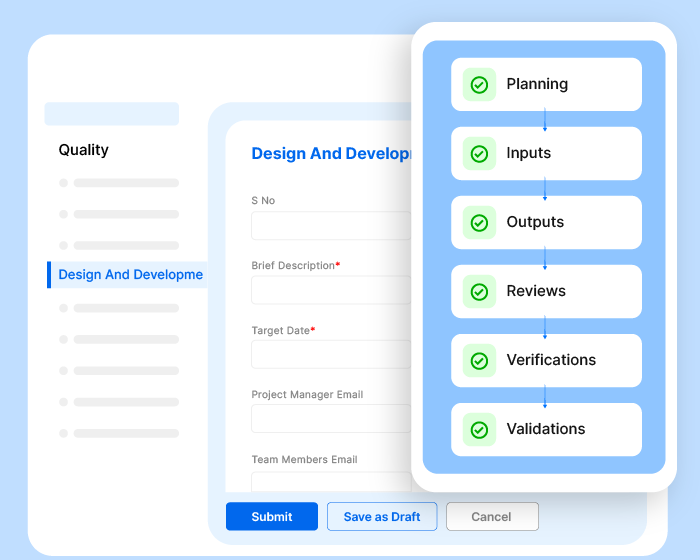
Effivity’s Quality Management Software is a unified platform for diverse needs. Tap into the power of an automated quality expert to manage governance, risks, compliance, documented information, internal audits audits, calibration, maintenance, customer complaint, training, objectives etc.
Experience efficiency like never before
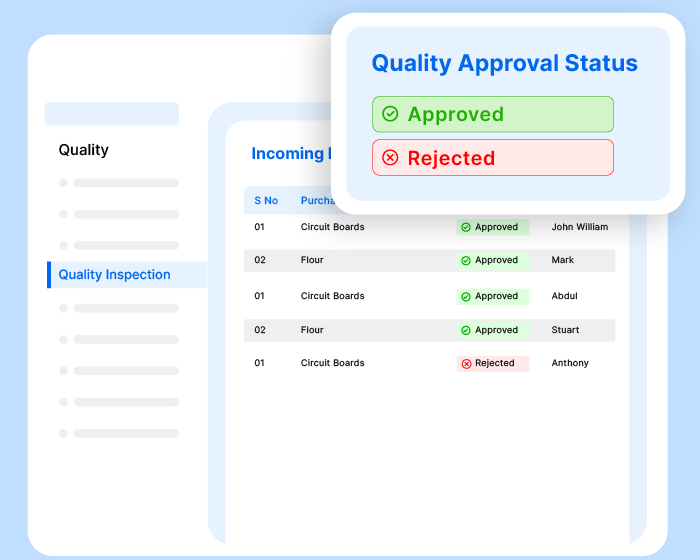
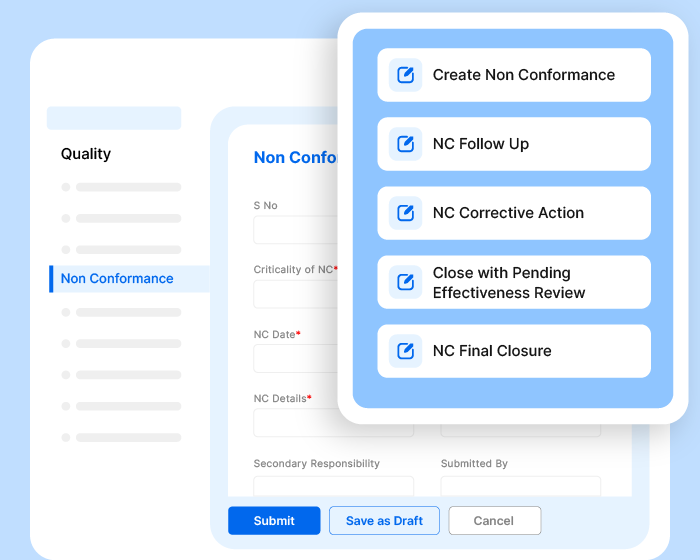
Conduct audits, approve documents, raise/close non-conformities, carry out inspections, track KPIs, assign tasks, automate workflows, generate records, and communicate with teammates - all effortlessly
Stay Connected,
Stay Successful
Maintain quality from anywhere in the world with our cloud-based quality management software. Access anything and everything via our mobile app remotely-from the field, your home, or even your favorite cafe!
Seamless Control at your Fingertips with 30+ integrations
Get all your tools in one place to maximize productivity. Connect and sync existing systems (third-party apps, CRM, ERP, etc.) with Effivity QMS and turbocharge your business
Ensure business continuity with Automated Backups
Keep your business up and running even after disruptions. Protect critical data, minimize downtime, and recover swiftly from disasters with automated data backups
100+ Pre-configured QMS Workflows
Access our library of more than 100 ready-to-use templates and workflows to get a 95% head start. Enjoy robust compliance and quality management from day one

Quality Management Brochure
Dive into Effivity ISO 9001 software modules, features and capabilities to unlock its true potential.
Compliance has never been this easy
Effivity QMS is a one-stop destination to build, certify, maintain, and enhance your quality compliance.
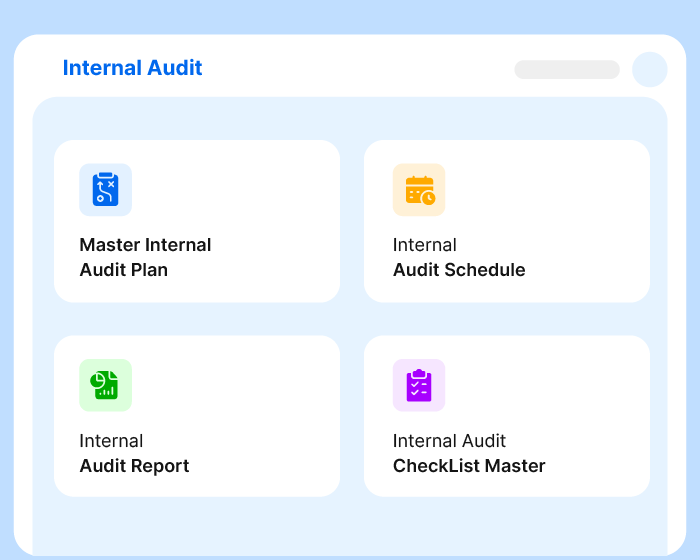
Schedule, Conduct, and Track Audits with Precision
Cut the audit-related admin hassle and get accurate results. Set trigger actions, monitor audit status, capture findings, and visualize insights to implement the best corrective measures.

A Single QMS Software for Multiple Quality Standards
Comply with ISO/IATF/AS/GMP/customer/ regulatory standards with a unified interface.

Automation and Reporting at its Best
Powerful reminders, notifications, and auto-escalation to complete tasks effectively and on time. Real-time dashboards with comprehensive data analytics and graphs further facilitate quick decision-making.

Implement & Maintain
ISO 9001 QMS with Effivity
Effivity’s Quality Management Software fits every business-small, medium, and large. We offer
numerous modules
covering different aspects of quality management. Pick your favorites, and
pay only for the features you want in the QMS!
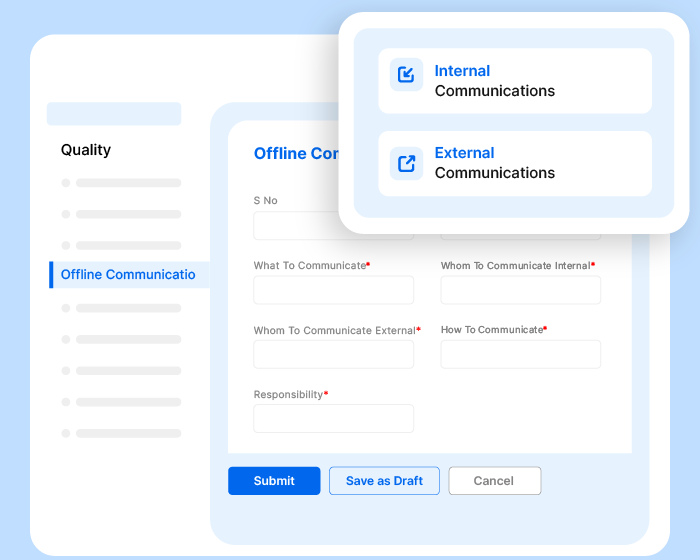
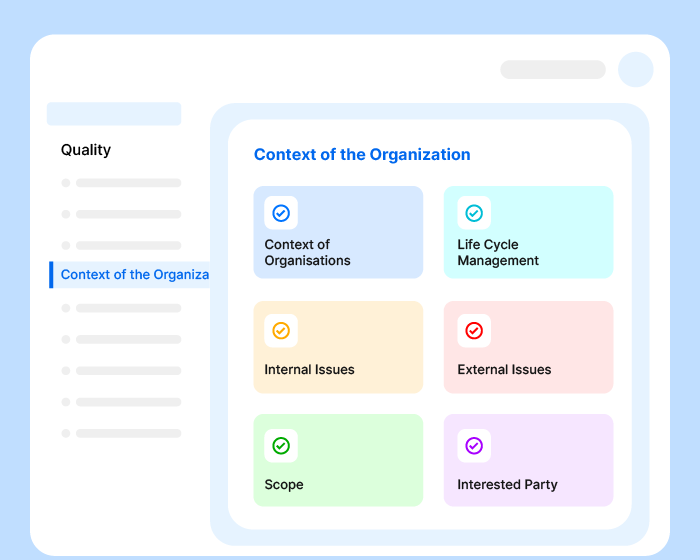
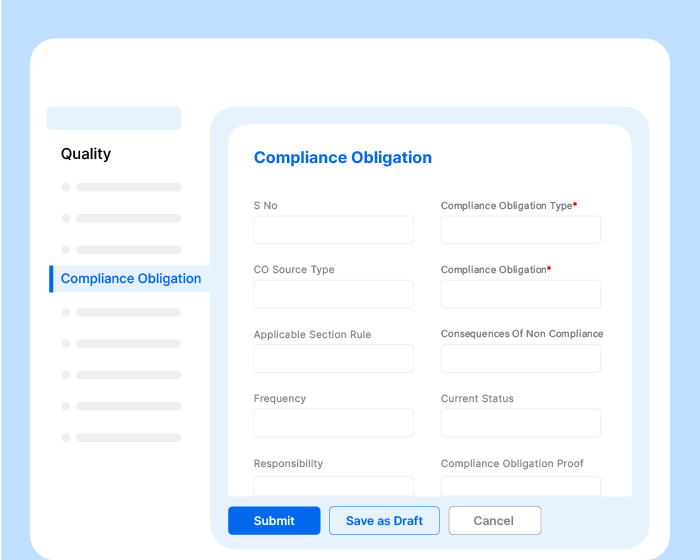
Effivity quality management solution allows you to define the communication process for your QMS, including internal and external communications.
Read more

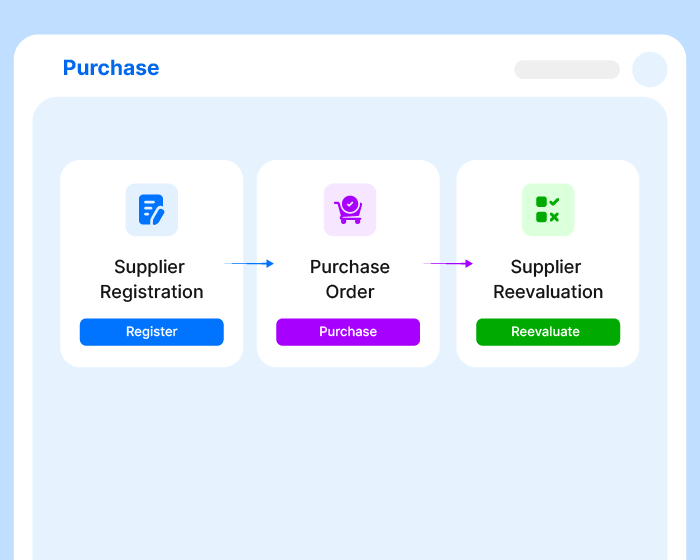
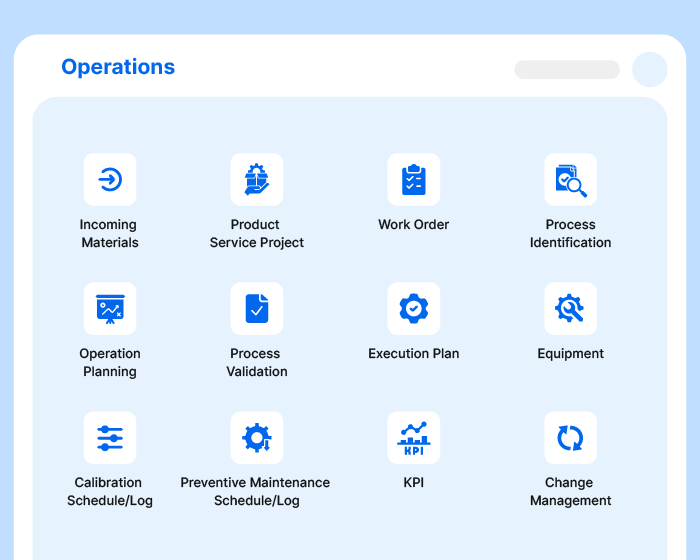
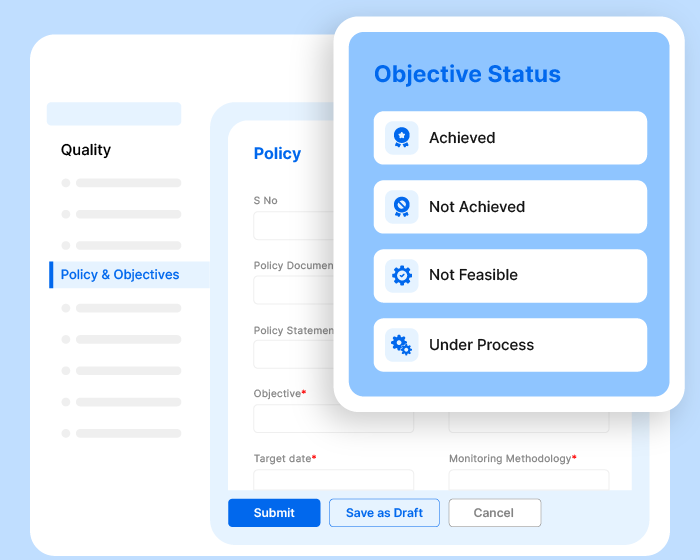
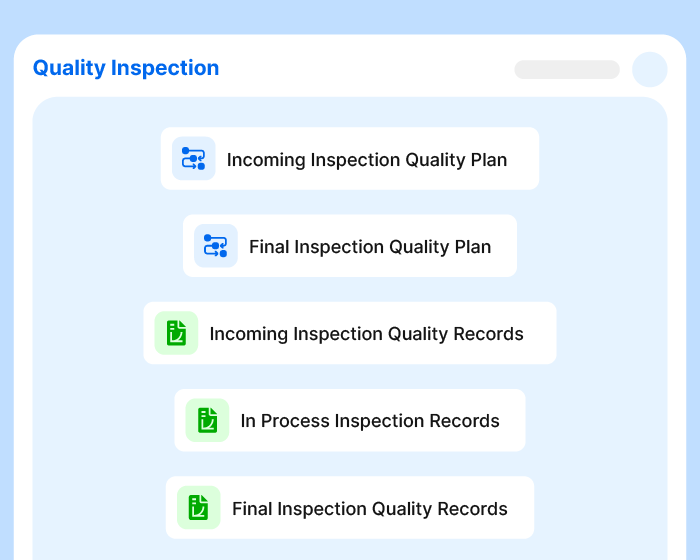


A Thousand Tasks.
A single tool
Effivity’s QMS software is an all-in-one platform for everything quality-products, services, and processes. Get it today to implement, maintain, and improve your quality management system.
Schedule a Free Demo
Why Choose Effivity
Quality Management Software?
Imagine a personal quality expert tailored to your business needs.
That’s exactly what
Effivity QMS is!
Redefine Quality with
Effivity QMS Automation
Free trials, quick deployment, and swift data migration. Achieve business excellence with stellar onboarding and customer support!
Get a free 2-week trial to explore our features. Add modules, build your subscription plan, and activate your subscription through bank transfer/ credit card.
Activate your account within seconds! Assign roles, configure access control, and even set up a multi-site structure.
Provide detailed training to admin and users on Effivity QMS. Our simple, intuitive interface and zero learning curve guarantee quick proficiency for all.
Roll out your QMS within a few days. Customize it to improve its efficiency and see the business performance skyrocket.
Phone, email, or online meeting. We’re always here to help you out.
Hear it from our customers
Don’t believe our word? Let our customers speak for our quality:
Great software that lays out all ISO requirements to fill in. It helps guide implementing an ISO system, not just a software to house documents/records.
Michelle F - Twist Bioscience
User friendly and innovative. 1. lessen my paper works 2. easily maintained company system. 3. It's updated as per the convenience of user. 4. easy system monitoring.
Lorraine B - Logistics and Supply Chain
I absolutely love this software. Gained a huge new customer in the market and were able to begin our ISO Certification journey.
Linda S. - Manufacturing Engineer
Enjoy limitless access:
anywhere, any screen, any time
Effivity QMS is responsive and device-independent. You can log in to your account via a web browser or download our app. PC, laptop, tablet or mobile phone-we’re everywhere.
Free Trial Today